Pathfinder
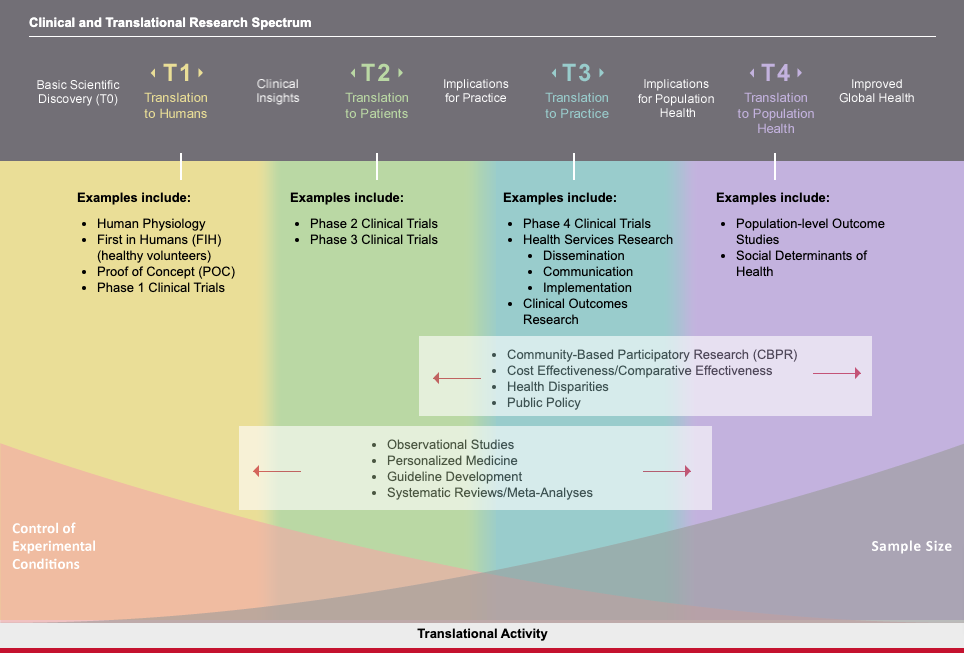
Clinical and translational research is characterized by a spectrum of activities where critical insights are passed between research modalities so that biomedical discoveries can lead to tangible improvements in human health.
References:
Pathfinder is based on material contained in the following three journal references.
Sung NS, Crowley WF Jr, Genel M, Salber P, Sandy L, Sherwood LM, et al. Central challenges facing the national clinical research enterprise. JAMA. 2003 Mar 12;289(10):1278-87. PubMed ID: 12633190
Westfall JM, Mold J, Fagnan L. Practice-based research – “Blue Highways” on the NIH roadmap. JAMA. 2007 Jan 24;297(4):403-6. PubMed ID: 17244837
Szilagyi PG. Translational research and pediatrics. Acad Pediatr. 2009 Mar-Apr;9(2):71-80. PubMed ID: 19329097

